What Drives Us
Our Mission
Providing leadership on rural priorities through advocacy, partnerships, and education.
Find Support
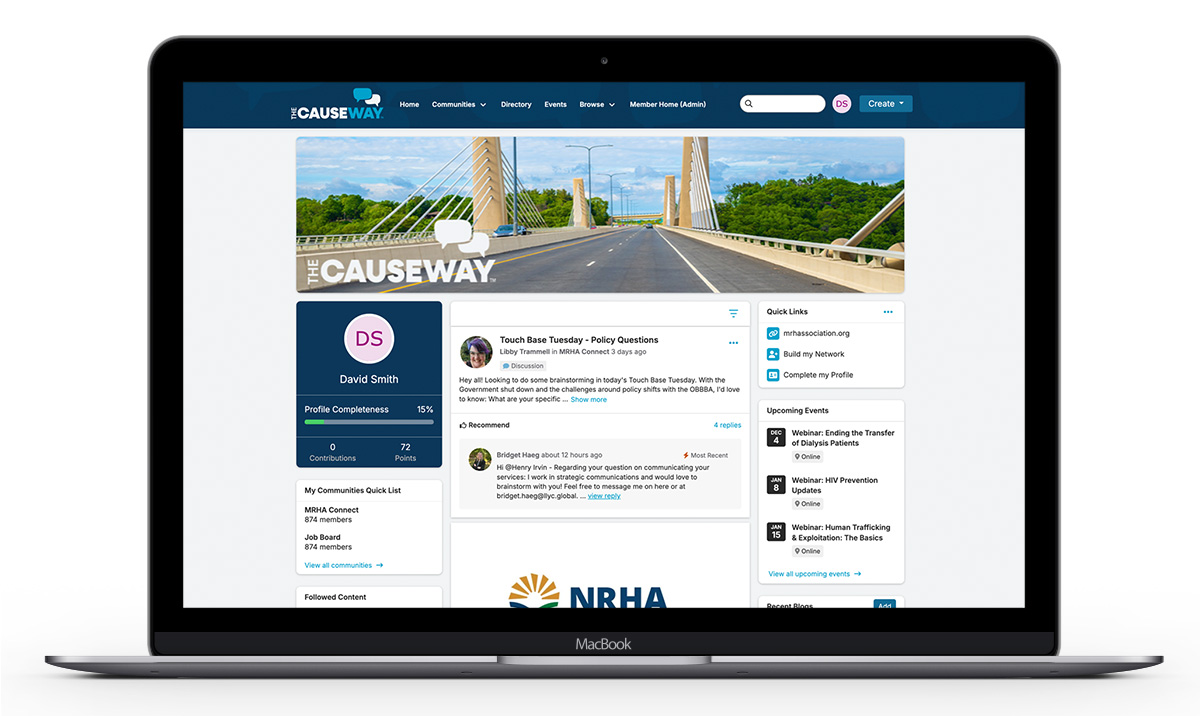
Join The Causeway- Missouri’s Bridge to Accessible Healthcare
The Causeway is Missouri’s shared platform where health advocates, executives, educators, and frontline caregivers come together to collaborate, share insights, solve challenges, and strengthen health outcomes statewide. Members gain access to shared resources, collective learning, partnership opportunities, and coordinated action that drive measurable progress toward accessible healthcare across Missouri’s communities.
Trainings and Events
From webinars and workshops to conferences and hands-on training, our events connect participants with experts and resources that empower them to improve healthcare delivery, stay informed on policy updates, and advance best practices.

